Uses of Acids and Bases
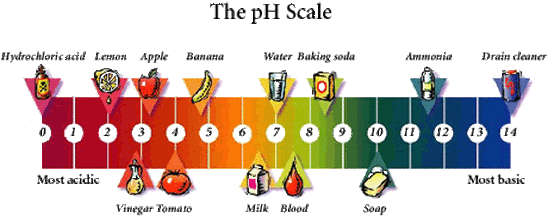
Below is a pH scale which you will be learning about soon, but for right now just focus on the types of things shown on the scale. Acids and bases are present in our everyday lives in the form of many things. Do you see that lemon? Well yes, lemons are classified as an acid, so you can say they are acidic. Do you see that leg? Well hair removal is a base, so you can say hair removal substances are basic.
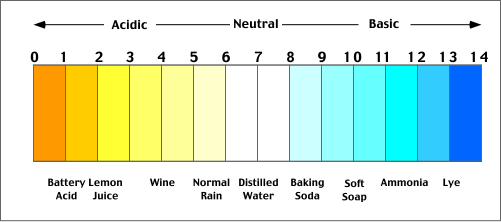
Acids and bases are used in our everyday lives without many of us knowing that they are actually acids or bases! Some of the many everyday acids and bases are shown in the chart below:
|
Acids:
|
Bases:
|
Other uses of acids include: benzoic acid(salt used to preserve food), carbonic acid(used to make carbonated drinks), ethanoic acid(used to clean metals before electroplating/processing leather/household cleaning/ maintenance of swimming pools), nitric acid(used in the production of fertilizers, explosives, dissolution of metals-extractiona and purification of gold), sulphuric acid(to make fertilizers, polymer, and detergent), and tartaric acid(manufacturing of sodas, gives tartness flavor to food, and used as a emetic- substance to make someone vomit).
Other uses of bases include: Ammonia(used in the production of fertilizers, used in manufacturing of nitric acid, to prevent clotting in natural and synthetic latex), aluminum hydroxide(often used to make gastric medicine-antacid), calcium hydroxide(used to make cement, limewater, help with application of sewage treatment, and neutralize soil's acidity), Sodium hydroxide(often used when manufacturing soaps, cleaners, and detergents), and magnesium hydroxide(used as an antacid, armpit deodorant that is antiperspirant, and non-hazardous alkali used to neutralize wastewater that is acidic).
Other uses of bases include: Ammonia(used in the production of fertilizers, used in manufacturing of nitric acid, to prevent clotting in natural and synthetic latex), aluminum hydroxide(often used to make gastric medicine-antacid), calcium hydroxide(used to make cement, limewater, help with application of sewage treatment, and neutralize soil's acidity), Sodium hydroxide(often used when manufacturing soaps, cleaners, and detergents), and magnesium hydroxide(used as an antacid, armpit deodorant that is antiperspirant, and non-hazardous alkali used to neutralize wastewater that is acidic).
How does the pH table work?
Well this picture above is a pH scale. It goes from 0-14, and gives you the range of what is considered acidic and what is considered basic. Any substance that is in the range of 0-6.9 is an acid, and everything from 7.1-14 is a base.
Girl you a ten and by that on the pH scale you basic girl.
Also here is a link to a periodic table of elements if you are in the need of one.
Also here is a link to a periodic table of elements if you are in the need of one.
Are you wondering what the properties of acids and bases are?
|
The properties of acids are as follows:
|
The properties of bases are as follows:
|
We have not learned some of the material in this property table, but do not worry, we will soon!
Below is a video of a strong base and acid reacting with aluminum coke cans:
Below is a video of a strong base and acid reacting with aluminum coke cans:
This can happen sometimes when eating acidic foods:
Q: Why do chemistry professors like to teach about ammonia?
A: Because it's basic stuff.