Conjugate Acid and base
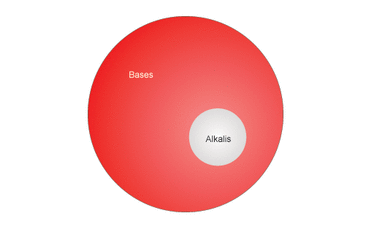
Well let's start off by defining the difference between an alkali and a base. An alkali is a base that dissolves in water. They can also be defined as a basic ionic salt from an alkali metal, or alkaline earth metal element. Many bases are insoluble (do not dissolve) in water, but if a base does, it is an alkali.
All alkalis are bases, but not all bases are alkalis (only soluble bases are).
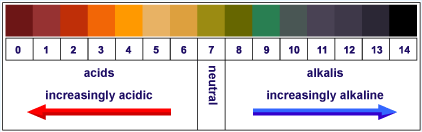
This pH scale says alkaline rather than basic, and now you know what the difference is.
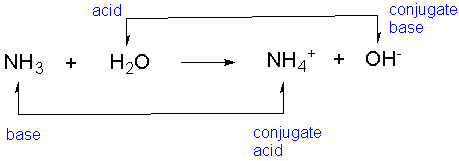
A Conjugate AcidThe substance formed after a base has received the proton (H+). It is the thing that gains a hydrogen.
|
A Conjugate BaseThe substance left over after the acid has lost its proton (H+).
|
- An example of a conjugate acid and base equation:
Can you guess which one is the acid, base, conjugate acid, and conjugate base?
Here is the answer