Strength of an Acid and Base
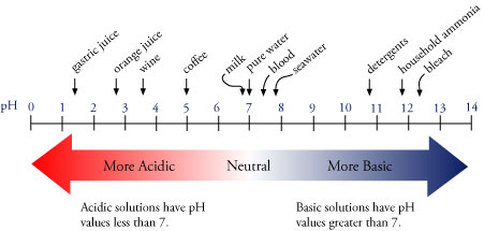
The closer you get to 0 on the pH scale, the more acidic a substance is, and the closer a substance is to 14, the more basic it is.
The definitions of a strong acid, weak acid, strong base, and weak base are as follows:
A strong acid=One that can completely dissociate to form hydrogen ions. Examples of strong acids are: hydrochloric acid, nitric acid, sulfuric acid, perchloric acid, chloric acid, hydrobromic acid, and hydroiodic acid.
A weak acid=One that partially dissociates, therefore cannot form hydrogen ions as readily. Examples of weak acids are: hydrofluoric acid and acetic acid.
A strong acid=One that can completely dissociate to form hydrogen ions. Examples of strong acids are: hydrochloric acid, nitric acid, sulfuric acid, perchloric acid, chloric acid, hydrobromic acid, and hydroiodic acid.
A weak acid=One that partially dissociates, therefore cannot form hydrogen ions as readily. Examples of weak acids are: hydrofluoric acid and acetic acid.
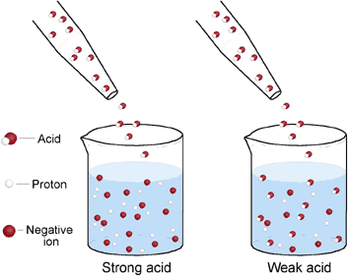
In this diagram you can see that the string acid all of the hydrogen ions (the protons) have dissociated (detached) themselves from the negative ions. In the weak acid you can see that not all of the hydrogen ions (protons) have dissociated though.
A strong base=One that dissociates easily to form hydroxide ions. Examples of a strong base are: sodium hydroxide, lithium hydroxide, potassium hydroxide, and rubidium hydroxide.
A weak base=Cannot dissociate as easily, therefore cannot form hydroxide ions as readily. Examples of weak bases are: ammonia and diethylamine.
A weak base=Cannot dissociate as easily, therefore cannot form hydroxide ions as readily. Examples of weak bases are: ammonia and diethylamine.
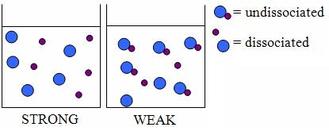
Here is another photo that demonstrates the link between dissociation and strength of an acid or base.
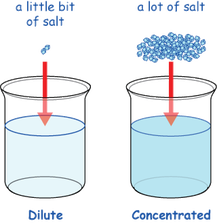
Concentration
Molarity= the amount of substance per unit volume
Concentration is based on the molarity of a substance. When strong acids and bases dissociate, they produce either hydrogen or hydroxide (depending on whether it is an acid or base), cause the concentration of those things to be high. As we already learned, the strong acids and bases produce more (hydrogen or hydroxide). If you added a strong acid or base to water, you would be diluting it, so the concentration of it would be low, but the substance would still be strong (as itself).
Concentration is based on the molarity of a substance. When strong acids and bases dissociate, they produce either hydrogen or hydroxide (depending on whether it is an acid or base), cause the concentration of those things to be high. As we already learned, the strong acids and bases produce more (hydrogen or hydroxide). If you added a strong acid or base to water, you would be diluting it, so the concentration of it would be low, but the substance would still be strong (as itself).