pH, pOH Concepts
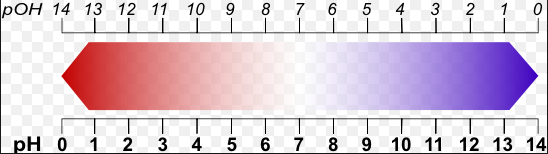
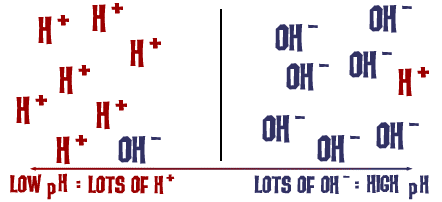
Still not sure what pH really means? pH is in short a measure of the basicity or acidity of a solution in which water is the solvent (aqueous solution). It is the measure of hydrogen ion concentration in a solution. The higher the concentration of hydrogen ions (H+), the lower the pH of a solution. If you have two acids with the same concentration of acid, the stronger acid would have a higher concentration of hydrogen ions than the weaker acid. So a stronger acid like hydrochloric acid has a low pH, while a weak acid like acetic acid.
pOH is the measure of the alkalinity of a solution, so basically it measures the hydroxide ions in a solution. It is the opposite of pH on the 0-14 scale because it says that a pOH of 14 is a strong acid, while a pOH of 0 is a strong base. The more hydroxide ions in a base the stronger it is.
pOH is the measure of the alkalinity of a solution, so basically it measures the hydroxide ions in a solution. It is the opposite of pH on the 0-14 scale because it says that a pOH of 14 is a strong acid, while a pOH of 0 is a strong base. The more hydroxide ions in a base the stronger it is.
So if there are a lot of pH ions the H is very low, while if there are a lot of OH- ions compared to H+ ions the pH is high.
[H+] stands for hydrogen ions, while [OH-] stands for hydroxide ions.
[H+] stands for hydrogen ions, while [OH-] stands for hydroxide ions.
How to calculate pH, pOH, [H+], and [OH-]
In order to understand how to calculate each of these things, you need to have formulas to do so.
The formulas to find each of these things are as follows:
pH= -log[H+]
pOH= -log[OH-]
pH+pOH=14
[H+]=2nd log{-pH]
[OH-]=2nd log[-pOH]
The formulas to find each of these things are as follows:
pH= -log[H+]
pOH= -log[OH-]
pH+pOH=14
[H+]=2nd log{-pH]
[OH-]=2nd log[-pOH]

It would be easier to do problems using these calculations with a calculator that looks like the one below, but all calculators that contain the functions needed in the formulas can be used.
Calculator is called a Ti -84 graphing calculator
Calculator is called a Ti -84 graphing calculator
Here lets do a practice problem:
So in the problem above, all you are given is pH. So go back to the equations above and look at which ones you could find using the pH. You can find the pOH by using the pH+pOH=14 formula, and you could find the [H+] using the [H+]=2nd log[-pH] formula.
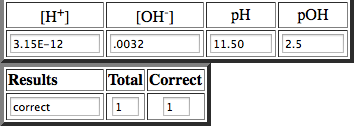
So we know the pH is 11.50, so we could subtract 11.50 from 14, to find the pOH. When you fill in the equation it looks like this, 14-11.50=2.5.
So the pOH=2.5.
We now have enough information to the the [OH-].
You just have to follow the formula, and plug in all of the information into your calculator correctly. You have to press 2nd and then log, and then type in negative whatever the pOH is that you have been given, or have already found.
[OH-]=2nd log[-pOH]
[OH-]=2nd log {-2.5]
[OH-]=.0032
To find the [H+], you can use this equation:
Here you do the same with the calculator, but instead of negative pOH, it is negative pH after you have pressed 2nd and then log.
[H+]=2nd log[-pH]
[H+]=2nd log[-11.50]
[H+]=3.15E-12
Answers may be slightly different past the decimal point due to rounding, but if you follow along, you will get a similar answer.
If you stick to the formulas though, you should get all of the correct answers.
So we know the pH is 11.50, so we could subtract 11.50 from 14, to find the pOH. When you fill in the equation it looks like this, 14-11.50=2.5.
So the pOH=2.5.
We now have enough information to the the [OH-].
You just have to follow the formula, and plug in all of the information into your calculator correctly. You have to press 2nd and then log, and then type in negative whatever the pOH is that you have been given, or have already found.
[OH-]=2nd log[-pOH]
[OH-]=2nd log {-2.5]
[OH-]=.0032
To find the [H+], you can use this equation:
Here you do the same with the calculator, but instead of negative pOH, it is negative pH after you have pressed 2nd and then log.
[H+]=2nd log[-pH]
[H+]=2nd log[-11.50]
[H+]=3.15E-12
Answers may be slightly different past the decimal point due to rounding, but if you follow along, you will get a similar answer.
If you stick to the formulas though, you should get all of the correct answers.
Do you feel like you are a mathematical genius now? If so, take the quiz.