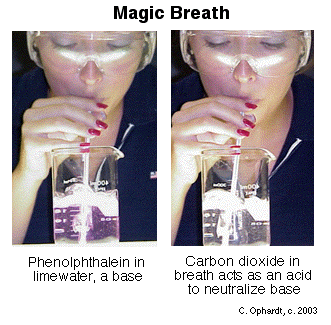
Neutralization of Acids and Bases:
Neutralization: is a chemical reaction where an acid and a base react to form a salt and water.
This type of reaction can be shown using this equation Acid+Base→Salt+Water
This type of reaction occurs when the amount of chemical substance or (moles) of a base and the amount of chemical substance or (moles) of an acid are equal. This causes the H+ ions that are from the acid to combine with the OH- ions that are from the base and create water, and that gets rid of the basic and acidic properties that the substances previously had. Salt is also created during this process, and the types will be specified in the next section.
This type of reaction can be shown using this equation Acid+Base→Salt+Water
This type of reaction occurs when the amount of chemical substance or (moles) of a base and the amount of chemical substance or (moles) of an acid are equal. This causes the H+ ions that are from the acid to combine with the OH- ions that are from the base and create water, and that gets rid of the basic and acidic properties that the substances previously had. Salt is also created during this process, and the types will be specified in the next section.
Ways to neutralize an acid:
-Put an acid with a metal
Acid +Metal→Salt +H2O
-Put an acid and an alkali together
Acid + Alkali→Salt +H2O
-Put an acid with a base
Acid +Base → Salt + H2O
-Put an acid with a carbonate
Acid + Carbonate→Salt + H2O +CO2
-Put an acid with a metal
Acid +Metal→Salt +H2O
-Put an acid and an alkali together
Acid + Alkali→Salt +H2O
-Put an acid with a base
Acid +Base → Salt + H2O
-Put an acid with a carbonate
Acid + Carbonate→Salt + H2O +CO2
Below is a video of a reaction between a strong acid and strong base:
It is an example of a neutralization reaction between hydrochloric acid and sodium hydroxide.
It is an example of a neutralization reaction between hydrochloric acid and sodium hydroxide.